KR Representative Service
Representative Services for Medical Devices in South Korea
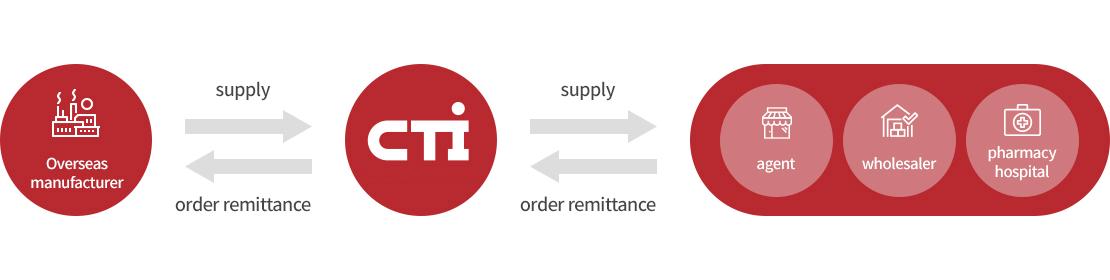
This is a service that performs the management of domestic product licenses and imports on behalf of foreign medical device manufacturers. Through this service, foreign manufacturers can obtain characteristics similar to those of a Korean branch through their legal representative without large initial investment costs, and domestic agents can focus on sales and marketing without going through difficult tasks such as product licensing and management.
The following services are available:
Item approvals and insurance registrations:
- import product approvals
- KGMP
- insurance registrations
- new medical technology evaluation
Import and customs clearance:
- order receipts and placing orders
- report and progress of scheduled customs clearance
- incoming and outgoing inspection
- incoming and outgoing records
- domestic quality controls
- warehouse rentals and inventory management
- collections and receivable management
Sales and marketing and follow-up management:
- management of nonconforming products
- report of side effects and corrective actions
- recalls
We promise the following four for companies aiming to enter the Korean market.
First, our accurate understanding of the Korean market will help you enter the Korean market quickly, allowing you to obtain domestic product licenses and discover domestic agencies at the same time. We also help you establish a domestic business model specialized for each product.
Second, several agencies and sales channels are secured by CTI will help you to expand and change agencies more freely when you enter the Korean market.
Third, securing business continuity maintains ownership of certification, and enables item permit management according to a high-level legal compliance system.
Fourth, you can get the effects such as reasonable service cost, service cost setting in consideration of business scale, import volume and profit, customized logistics cost proposal, cost-effective domestic branch agency, reduction of initial infrastructure investment required when establishing a Korean branch.

